Research Overview
The discoveries that specialized cells can be reprogrammed into other differentiated cell types and into pluripotent stem cells have revolutionized stem cell research.
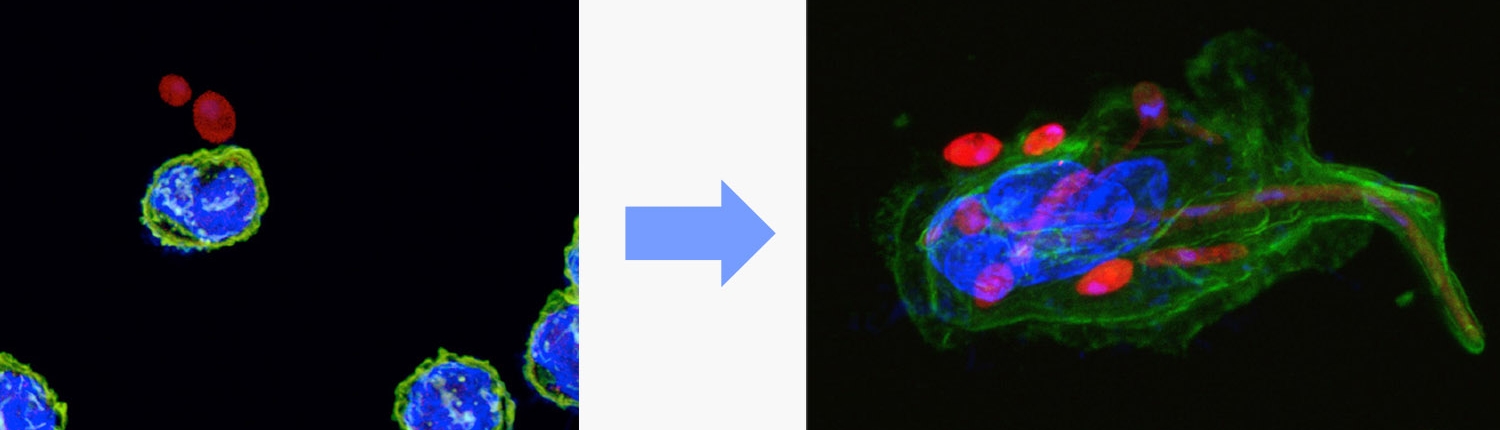
B cell to macrophage transdifferentiation
Our laboratory has pioneered the transcription factor-induced transdifferentiation of blood cells, originally with cells from chickens and in the last 1.5 decades with mouse and human cells. In particular, we have succeeded in converting mouse B cells into functional macrophages by the enforced expression of C/EBPa, a transcription factor produced by macrophages and other cell types. This process is 100% efficient and requires only about 3 days (see video). Using an inducible form of the transcription factor we also established a human B cell leukemia cell line that can be converted into non-tumorigenic monocytes. These cells have become a popular model to study inflammatory processes.
Cell reprogramming into iPS cells
More recently we found that C/EPBa also plays an important role in the reprogramming of B cells into pluripotent cells (iPSCs) by the Yamanaka factors Oct4, Sox2, Klf4 and Myc (OSKM). Thus, exposing pre-B cells to a pulse of C/EBPα followed by OSKM expression induces their reprogramming into iPS cells in nearly all cells. During this conversion a substantial proportion of the cells express endogenous Oct4 within 2 days (see figure) and undergo an epithelial-mesenchymal transition. The process is initiated by the C/EBPα-induced activation of the dioxygenase Tet2, whose overexpression on its own enhances the OSKM-induced reprogramming efficiency. Tet2 modifies methylated cytosines and facilitates their complete demethylation, thus relieving gene repression. This provides a mechanistic link between induced B-cell transdifferentiation and iPS cell reprogramming. The C/EBPa pulse also mediates an increased accessibility of pluripotency genes to Oct4 binding by as yet poorly understood mechanisms. Our discovery that the sequence specific transcription factor C/EBPa can poise B cells to rapid and efficient reprogramming by the Yamanaka factors into pluripotent cells may reflect its possible role in early embryogenesis, another topic that we are investigating.
Investigating molecular mechanisms
The uniquely efficient transdifferentiation and iPSC reprogramming systems developed allow to study the underlying molecular mechanisms with unparalleled precision. Using these mouse and human models our current work centers on the effects of cell fate-instructive transcription factors on genome topology, methylation, chromatin accessibility and epigenetics.